Skin-interfaced wireless platform for intrapartum fetal and maternal monitoring
Intrapartum fetal monitoring has remained largely unchanged for nearly 50 years, with Electronic Fetal Monitoring introduced in the 1970s to identify fetal asphyxia and prevent related brain injuries like hypoxic-ischemic encephalopathy [1]. Despite its introduction, the rate of Cesarean sections (C-sections) in the U.S. has increased fivefold, while rates of birth asphyxia-related conditions like cerebral palsy have not decreased. The high rate of false positives in cardiotocography-based monitoring often leads to unnecessary interventions, including “emergency” C-sections, a concern recognized by organizations such as the American College of Obstetricians and Gynecologists [2], [3].
To address this, we propose developing an advanced, integrated monitoring platform that uses flexible electronics, wireless connectivity, and compatibility with low-cost mobile devices. This platform will provide comprehensive, time-synchronized monitoring of both maternal and fetal vital signs, including non-invasive maternal and fetal heart rate, and uterine contraction monitoring. The platform will be evaluated in a term-pregnant ewe model undergoing induced labor, laying the groundwork for future clinical applications and potentially reducing unnecessary C-sections by improving intrapartum fetal monitoring.
Vital signs monitoring is essential for ensuring the health and safety of women and newborns during pregnancy, labor, and childbirth. It is often the first step in detecting pregnancy abnormalities, providing opportunities for timely interventions to prevent maternal and neonatal morbidity and mortality. Contemporary pregnancy monitoring systems require multiple wired devices connected to large base units, which are often complex, expensive, and challenging to implement in low-resource settings where maternal morbidity and mortality are greatest. This project will make foundational technical contributions by: (1) developing skin-interfaced flexible electronic tattoos (e-tattoos) that attach to the abdomen for continuous monitoring of materal cardiac activity and uterine contraction, and fetal cardiac activity during labor and delivery; (2) investigating novel machine learning algorithms to extract effective data representations from mixed multi-channel maternal-fetal electrophysiological data, with a focus on detecting changes in fetal heart rate; and (3) conducting a comprehensive evaluation of the technology through a field study in low-resource and high-resource settings.
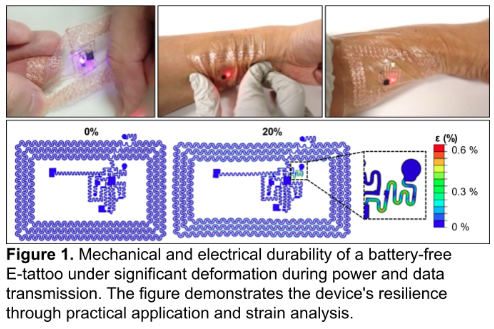
E-tattoos are ultrathin (approximately 100 µm excluding ICs) and ultrasoft membranes that conform to the skin, enabling the monitoring of various biomarkers, including ECG, mechano-acoustic signals, skin temperature, skin hydration, skin stiffness, and maternal blood oxygen saturation (Figure 1) [4]. These e-tattoos integrate Near Field Communication (NFC) and Bluetooth Low Energy (BLE) technologies for wireless power delivery, configuration, and data transmission. The form factor provides soft mechanical characteristics for comfortable skin adhesion, even under mechanical stress in sensitive body regions. Experimental studies and finite element analysis (FEA) confirm that the copper in the serpentine interconnects (250 µm width) remains below fracture limits (ε = 0.6%) during operation under various external forces, ensuring the multilayer e-tattoo can survive skin-tolerable deformation (up to 20%) without mechanical or electrical failure. These characteristics guarantee soft, low-irritation interfaces with the skin, even when extreme curvatures are encountered.
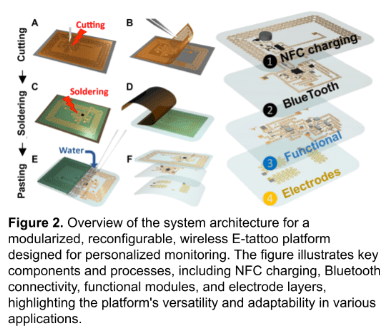
The NFC and Bluetooth-enabled e-tattoo (Figure 2) is designed with modular and reconfigurable fabrication, consisting of four layers: NFC, Bluetooth, functional, and electrode layers. The wireless e-tattoo can be fully powered by an interrogator, with rectified power used for an optional rechargeable coin cell battery, as well as powering the Bluetooth system-on-chip and multiple active components, including an Analog Front End (AFE) IC and Inertial Measurement Unit (IMU). This design emphasizes reconfigurability and reusability, allowing the e-tattoo to include reusable and replaceable electronic modules or disposable electrode modules, based on user needs. Each layer represents a separate electric circuit, supported by transparent medical tape with adhesive on one side. Through optimized design and characterization, we have demonstrated multiple sensing modalities with this battery-free, wireless e-tattoo, including electrocardiogram (ECG) Electromyography (EMG), oxygen saturation (SpO2), heart rate, skin temperature, and skin hydration. The optimized choice of tracked biomarkers will depend on the personal needs and specific situation of the user [5], [6], [7], along with a closed-loop feature [8], [9].
In my current work, we developed a wearable network system for monitoring several maternal and fetal vital signs (Figure 3). This platform contains flexible, soft, and low-profile sensors, allowing for longitudinal assessment of parameters like continuous maternal/fetal HRs (MHR, FHR) and EMG-derived uterine activity (UA) monitoring. The system integrates with the cloud, enabling the development of data analytics to predict maternal or neonatal outcomes. The platform utilizes a thin, flexible printed circuit board in an open architecture, with elastomeric encapsulation to seal the system and facilitate sterilization for re-use. The layouts ensure soft mechanical characteristics for comfortable attachment to the abdomen, with a touch-free docking interrogator for wireless charging and automated data downloads to minimize user burden. The user interface transmits data to a cloud hub for processing and extraction of clinical-grade MHR, FHR, and UA, graphically displayed in an intuitive manner. This automated system is particularly important for longitudinal validation in human subjects.
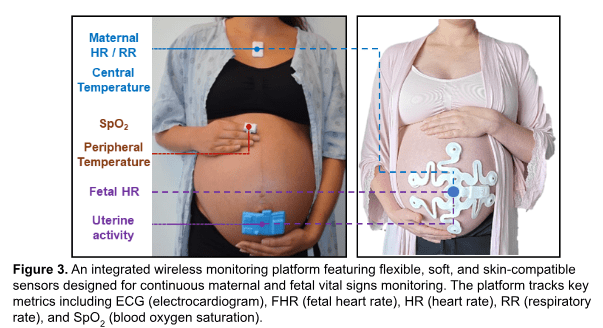
The system’s performance was recently validated in both low- and high-resource settings. Successful field trials were conducted among pregnant women between 25 and 41 weeks of gestation (n = 40) in both low-resource settings (Zambia and Gana) and high-resource settings (Chicago, IL). The proposed project aims to address a widespread health issue that impacts nearly every family, with the potential to improve childbirth outcomes and reduce associated costs.
References
[1] R. K. Freeman, “Problems with intrapartum fetal heart rate monitoring interpretation and patient management1,” Obstetrics & Gynecology, vol. 100, no. 4, pp. 813–826, Oct. 2002, doi: 10.1016/S0029-7844(02)02211-1.
[2] K. B. Nelson, T. P. Sartwelle, and D. J. Rouse, “Electronic fetal monitoring, cerebral palsy, and caesarean section: assumptions versus evidence,” BMJ, p. i6405, Dec. 2016, doi: 10.1136/bmj.i6405.
[3] Z. Alfirevic, G. M. Gyte, A. Cuthbert, and D. Devane, “Continuous cardiotocography (CTG) as a form of electronic fetal monitoring (EFM) for fetal assessment during labour,” Cochrane Database of Systematic Reviews, vol. 2019, no. 5, Feb. 2017, doi: 10.1002/14651858.CD006066.pub3.
[4] H. Jeong et al., “Modular and Reconfigurable Wireless E‐Tattoos for Personalized Sensing,” Adv. Mater. Technol., vol. 4, no. 8, p. 1900117, Aug. 2019, doi: 10.1002/admt.201900117.
[5] H. Jeong, J. A. Rogers, and S. Xu, “Continuous on-body sensing for the COVID-19 pandemic: Gaps and opportunities,” Sci. Adv., vol. 6, no. 36, p. eabd4794, Sep. 2020, doi: 10.1126/sciadv.abd4794.
[6] H. Jeong et al., “Differential cardiopulmonary monitoring system for artifact-canceled physiological tracking of athletes, workers, and COVID-19 patients,” Sci. Adv., vol. 7, no. 20, p. eabg3092, May 2021, doi: 10.1126/sciadv.abg3092.
[7] H. Jeong et al., “Miniaturized wireless, skin-integrated sensor networks for quantifying full-body movement behaviors and vital signs in infants,” Proc. Natl. Acad. Sci. U.S.A., vol. 118, no. 43, p. e2104925118, Oct. 2021, doi: 10.1073/pnas.2104925118.
[8] Y. S. Choi, H. Jeong et al., “A transient, closed-loop network of wireless, body-integrated devices for autonomous electrotherapy,” Science, vol. 376, no. 6596, pp. 1006–1012, May 2022, doi: 10.1126/science.abm1703.
[9] H. Jeong et al., “Closed-loop network of skin-interfaced wireless devices for quantifying vocal fatigue and providing user feedback,” Proc. Natl. Acad. Sci. U.S.A., vol. 120, no. 9, p. e2219394120, Feb. 2023, doi: 10.1073/pnas.2219394120.
Reported by Dr. Hyoyoung Jeong (2023 KSEA YIG Grant Recipient)
